Clinical Practice Guidelines · May 3, 2026
Applying Clinical Guideline Endorsement Principles to Innovator Visa Success
Learn how the rigour of clinical practice guideline endorsements can inform your Innovator Visa application process, enhanced by Torly.ai’s AI-driven insights.

Why “Clinical Guidelines Endorsement” Holds the Key to Visa Triumph
Think of clinical guidelines endorsement in oncology: every recommendation is backed by solid evidence, expert review and constant updates. Now imagine applying the same rigour to your UK Innovator Visa application. You shift from guesswork to a structured, data-driven approach.
In practice, this means treating your business plan like a clinical protocol—every claim verified, every risk assessed, every step peer-reviewed. It sounds intense but it need not be daunting. With the right AI-driven tools you gain clarity, confidence and a clear roadmap. Boost your clinical guidelines endorsement with our AI-Powered UK Innovator Visa Application Assistant
By mirroring the precision of medical guidelines, you avoid common pitfalls: missing evidence, vague goals or poorly justified projections. The result? A stronger pitch to endorsing bodies and a faster route to visa success.
From Oncology Protocols to Visa Pathways: Why Rigour Matters
Clinical practice guidelines in cancer care are the gold standard. They follow a strict process:
- Systematic literature reviews
- Graded recommendations
- Multi-stakeholder consensus
- Regular revisions
Each recommendation is debated by experts. Conflicts are declared. Patient safety and outcomes drive decisions.
Now compare that to many visa applications: unsubstantiated claims, incomplete financial forecasts, or cookie-cutter business plans. You know the drill—yet it still happens.
By adopting guideline-style rigour you:
- Build trust with endorsing bodies
- Anticipate their questions
- Address concerns before they arise
Ultimately, you create an endorsement-ready application, not just a hopeful submission.
Three Pillars of Clinical Guideline Endorsement Adapted for Visa Success
-
Evidence-Based Business Idea Qualification
– In medicine, every recommendation is graded by evidence level.
– For your visa, back each assertion with market data, pilot results or technical proof.
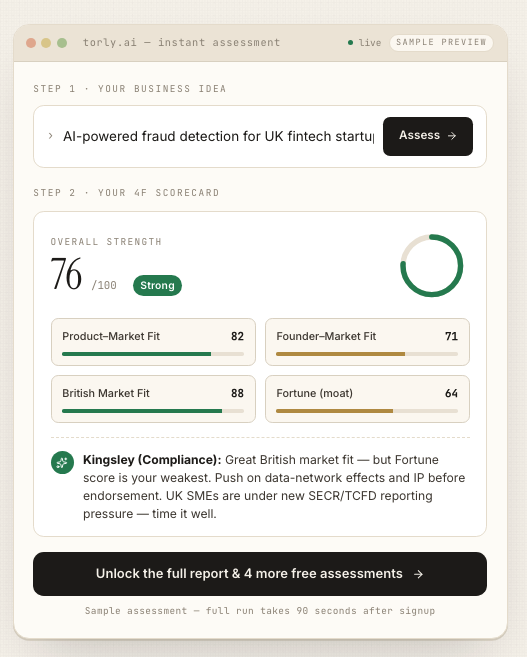
– Torly.ai’s Business Idea Qualification agent evaluates innovation, feasibility and scalability against Home Office criteria. -
Individualised Applicant Assessment
– Just as clinicians tailor treatment by patient history, you personalise your pitch by highlighting your expertise and track record.
– Torly.ai’s Background Assessment agent analyses your CV, projects and achievements to ensure they align with endorsing body standards. -
Continuous Feedback and Iteration
– Oncology guidelines evolve with new research.
– Torly.ai’s Gap Identification & Action Roadmap agent pinpoints weaknesses, then suggests precise improvements—whether refining your financial model or strengthening your IP strategy.
After you review feedback, you refine your plan. Then you repeat the cycle. Over time your application becomes bullet-proof.
Feeling ready to structure your venture like a clinical protocol? Build your Business Plan NOW with TorlyAI Desktop APP
Case Study: Navigating Antibiotic Stewardship Lessons for Visa Endorsement
The Infectious Diseases Society of America (IDSA) recently refused to endorse certain pneumonia guidelines, citing risks of indiscriminate antibiotic use in viral cases. They argued for dynamic, patient-centred decisions rather than blanket protocols.
Lessons for visa applicants:
- Avoid one-size-fits-all pitches
- Anticipate objections (the “viral assay” of finance projections)
- Briefly pause to gather more evidence if needed
Imagine you propose an ambitious tech solution. Endorsing bodies may question your market research or revenue assumptions. If you rush, you risk rejection. Instead, follow the IDSA approach:
- Gather precise data on customer adoption rates
- Reassess projections once new metrics arrive
- Document every change
Torly.ai helps you mimic this dynamic decision-making. Its real-time feedback engine flags potential issues so you can pause, investigate and refine—just like prudent antibiotic stewardship.
Actionable Steps to Streamline Your Innovator Visa Application
Here’s your checklist, inspired by clinical guideline development:
• Conduct a systematic market review
– List primary sources (industry reports, competitor analysis)
– Grade each insight by reliability
• Draft your initial business plan
– Clearly define problem, solution, target market
– Include evidence (pilot data, letters of intent)
• Submit to Torly.ai for multi-layer assessment
– Business idea, applicant background, compliance checks
• Review the Gap Identification report
– Prioritise recommendations (technical, financial, operational)
– Track changes in a revision log
• Iterate until your plan earns a high endorsement likelihood score
With this disciplined workflow, you transform your application from rough draft to polished submission in under two weeks on average.
Halfway through and eager to test these steps? Ready to apply clinical guidelines endorsement to your visa strategy? Try our AI-Powered UK Innovator Visa Application Assistant
Why Torly.ai Outshines Traditional Consultancy
Sure, you could engage a visa coach or fill out forms manually. Names like VisaHQ and SimpleVisa promise support. They do a decent job organising paperwork. But what they lack is:
- Continuous, real-time reasoning
- Automated gap-analysis across business, finance and compliance
- Instant updates aligned with evolving Home Office rules
Torly.ai stands out because it:
- Provides 24/7 AI support so you never wait for office hours
- Generates tailored business documentation that meets endorsing body criteria
- Boasts a 95% success rate based on historic application outcomes
- Delivers feedback and a full action roadmap in under 48 hours
In short, Torly.ai blends deep business analysis with immigration expertise, offering a seamless, user-friendly route to endorsement.
Need a hands-on tool to build your endorsement application? Create your endorsement-ready business plan with 6 AI agents
Testimonials
“Torly.ai guided me step by step, just like a clinical trial protocol. I had all my gaps identified and fixed in days. I’m now in the UK with my Innovator Visa.”
– Priya S., Tech Entrepreneur
“I loved how Torly.ai’s AI agents break down complex requirements. The dynamic feedback reminded me of peer review in medicine.”
– David K., Biotech Founder
“From raw idea to endorsement-ready plan in under a week. It felt like having a 24/7 expert consultant on call.”
– Aisha M., HealthTech CEO
Conclusion
Adopting clinical guidelines endorsement principles transforms your visa application process. You move from uncertainty to confidence, guided by robust evidence, continuous feedback and expert-level analysis. Whether you’re a seasoned founder or a first-time innovator, this approach ensures your application is endorsement-ready.
Take control of your Innovator Visa journey today. Experience the power of clinical guidelines endorsement with our AI-Powered UK Innovator Visa Application Assistant